DNA binding proteins interact with DNA by means of various structural motifs, and can stimulate or repress transcription of messenger RNA, depending on the properties and location of the DNA sequence to which they bind.
DNA binding proteins are classified into four types:- homeodomain proteins, zinc finger proteins, leucine zipper proteins, and helix loop helix proteins.
I) Homeodomain protein :
a) Octanucleotide binding protein 1 (OCT –1), which regulates the histone gene H2B, the thymine kinase gene, and SnRNP genes
b) Octanucleotide binding protein 2 (OCT-2) , which regulates various immunoglobulin genes.
pituitary specific factor 1(Pit 1), which regulates the growth hormone gene, the thyroid stimulated hormone gene, and the prolactin gene.
II) Zinc finger protein :
DNA binding proteins are classified into four types:- homeodomain proteins, zinc finger proteins, leucine zipper proteins, and helix loop helix proteins.
I) Homeodomain protein :
- This protein consists of three linked alpha helices (helices 1, 2and 3). Helices 2 and 3 are arranged in a conspicuous helix turn helix motif.
- A 60 amino acid long region (homeodomain) within helix 3 binds specifically to DNA segments that contain the sequence 5’ATTA3’
a) Octanucleotide binding protein 1 (OCT –1), which regulates the histone gene H2B, the thymine kinase gene, and SnRNP genes
b) Octanucleotide binding protein 2 (OCT-2) , which regulates various immunoglobulin genes.
pituitary specific factor 1(Pit 1), which regulates the growth hormone gene, the thyroid stimulated hormone gene, and the prolactin gene.
II) Zinc finger protein :
Two types of zinc finger have been reported, called C2H2 finger and C4 finger
C2H2 zinc finger is a loop of 12 amino acids with two cysteines and two histidines at the base of the loop that tetrahedrally coordinate a zinc ion.
C4 finger
a) it has one alpha helix
b) It contains one zinc atom bound to four cysteine amino acids
c) It contains a hormone binding region
d) A 70 amino acid long region near the zinc atom binds specifically to DNA segments.
a) it has one alpha helix
b) It contains one zinc atom bound to four cysteine amino acids
c) It contains a hormone binding region
d) A 70 amino acid long region near the zinc atom binds specifically to DNA segments.
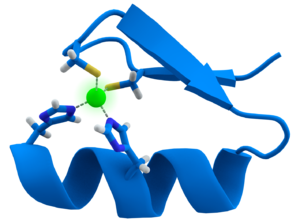
Examples of Zinc finger proteins are:
a) Transcription factor IIIA(TFIIIA), which engages RNA polymerase II to the gene promoter
b) Sp1(First described for its action on the SV40 promoter), which engages RNA polymerase II to the gene promoter by binding to the GC box
c) Glucocorticoid receptor, estrogen receptor, progesterone receptor, thyroid hormone receptor (erbA), retinoid acid receptor, and the vitamin D3 receptor.
III) Leucine Zipper Protein
- It consists of of an alpha helix that contains a region in which every seventh amino acid is leucine, which has the effect of lining up all the leucine residues on one side of the alpha helix.
- The leucine residues allow for dimerization of the two lecine zipper proteins and formation of Y shaped dimer.
- Dimerization may occur between two of the same proteins(homodimers, eg., Jun-Jun) or two different proteins(heterodimers, eg., Fos-Jun).
- It contains a 20 amino acid long region that binds specifically to DNA segments.
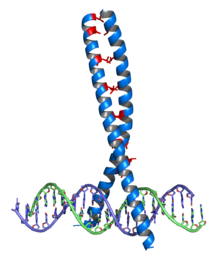
Example of Leucine Zipper proteins are:
a)CCAAT/ enhancer binding protein (C/EBP), which regulates the albumin gene and the alpha 1 anti trypsin gene.
b) Cyclic AMP response element binding protein (CREB), which regulates the somatostatin gene and the proenkephalin gene.
c)Finkel osteogeneic sarcoma virus (Fos) protein, a product of of the c fos protooncogene, which regulates various genes involved in the cell cycle and cell transformation.
d)Jun Protein, a product of the c-jun proto-oncogene, which regulates various genes involved in the cell cycle and cell transformation.
IV) Helix loop helix protein (HLH)
- It consists of a short alpha helix connected by a loop to a loner alpha helix.
- The loop allow for dimerization of two HLH proteins and formation of Y shaped dimer.
- Dimerization may occur between two of the same proteins (homo dimers) or two different proteins (heterodimers)
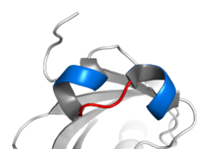
Example of Helix loop helix proteins are:
a) MyoD protein, which regulates various gees involved in muscle development.
b) Myc protein, a product of the c myc protooncogene, which regulates various genes involved in the cell cycle.
Tags:
DNA binding domains
example of helix loop helix.
example of homeodomain example of zinc finger
example of leucine zipper
leucine zipper proteins
Types of DNA binding proteins
zinc finger proteins

Thank you
ReplyDelete